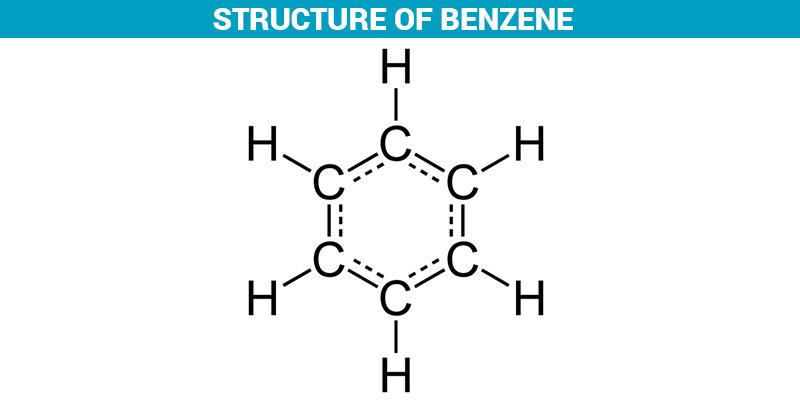
Nonpolar covalent bonds are a kind of bond that occurs when two atoms share a couple of electrons with each other. Some types of chemical bonding are very comparable to the way that children play with toys. Have you ever saw toddlers playing together with a toy? Sometimes they uniformly share toys, and other times, one child takes the other child’s toy away. In a nonpolar covalent bond, the atoms share electrons evenly with one another. The covalent bond can be of two kinds, specifically, polar covalent bonds and non-polar covalent bonds. Here, both the atoms partake an electron to form a bond. In this variety of bonding, the electrons in the atoms of the molecules are shared between each other. A positively energized ion is called a cation, and a negatively energized ion is called an anion.Ĭovalent bonding: It is one of the varieties of chemical bonding. Therefore, the atom forms ions, i.e, a cation, and an anion. Here, one atom gives the electron and the other atom accepts the electron to create a bond. In this variety of bonding, the electrons in the atoms of the molecules are transferred between each other. Ionic bonding: It is one of the types of chemical bonding. The chemical bond that occurs between molecules can be of two types, namely, ionic and covalent bonds. 
Chemical Bonding: Polar and Nonpolar Covalent BondsĪ bond is a chemical affinity between two atoms in which the electrons staying in an atom make a bond between two atoms. Then, you can dissolve the ethanol solution into an organic solvent, such as xylene. For example, if you want to mix an ionic compound or polar compound in an organic solvent, you may be capable to dissolve it in ethanol (polar, but not by a lot). It’s valuable to know which compounds are halfway between polar and nonpolar because you can use them as an intermediate to dissolve a chemical into one it wouldn’t associate with otherwise. This is why oil and water don’t associate: oil is nonpolar while water is polar. The general law is that “like dissolves like”, which indicates polar molecules will dissolve into other polar liquids and nonpolar molecules will dissolve into nonpolar liquids. If you know the duality of molecules, you can foretell whether or not they will associate together to form chemical solutions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
